
Most poisoning accidents involve medicines, household products and cosmetics. According to the World Health Organization (WHO), child-resistant packaging is one of the best-documented successes in preventing the unintentional poisoning of children. It also observed that medicinal drugs are the leading cause of non-fatal poisoning in children in middle and high income countries and in the USA.
Child-resistant (CR) packaging is used to reduce the risk of poisoning in children via the ingestion of potentially hazardous items including certain prescription and over-the-counter (OTC) medications, pesticides, and household chemicals.
According to the US Poison Prevention Packaging Act (PPPA), the term “Child-resistant” means that packaging is designed or constructed to be significantly difficult for children under five years of age to open or obtain a toxic or harmful amount of the substance contained therein within a reasonable time and not difficult for normal adults to use properly.

It doesn’t mean that children from this category cannot open a CR-packaging within a reasonable time. The CR-packaging is intended to be a last line of defence with safe and appropriate storage of medicines being the primary preventative measure in harm reduction.
In the early 1970s when the first CR-packaging appeared on the market after the PPPA had published its direction, there only was one packaging format (the bottle or jar) used in this market segment. For pharmaceutical and medicinal products blister packs or original pack dispensing (OPD) packs were uncommon, and consequently we saw the transformation of the bottle cap into a child-resistant closure (CR) as the singular approach to this problem.

But today we have a much more diversified market for medicines as now more than 50% of prescription drugs are dispensed in blister packs and potentially hazardous household products often are packaged in aerosols or spray bottles, representing a potential danger of child poisoning.
The blister pack is an excellent packaging format for medicines and a range of other products, as it incorporates a perfect seal, preventing contact either with the atmosphere or other products and permits dispensing with no risk of contamination at the retail level.
Similar arguments can be used to describe the popularity of aerosols and spray bottles in the household market, and also here, thus far, the relevant consumer goods companies have only toyed with CR-blisters and CR-sprayers. The prescription drugs market as well as the household chemicals market is often exclusively serviced with containers (bottles and jars) with the traditional and trusted CR-cap closure.

Burgopak’s child-resistant blister pack uses a pressure point locking system. Read about this design in my article “Developments In Pharmaceutical Packaging”. To read the article click the image.
The CR-closure for bottles and jars is well known, so I made a selection of recent developments in CR-packaging outside the bottle and jar sphere. I have 3 CR-designs for aerosols and/or spray bottles, 2 recent launches of CR-folding cartons with blisters, a CR-solution for flexible (stand-up) pouches and a CR-blister lidding option.
It will be a long story, so the 3 CR-designs for aerosols go to the next article. In this one the folding cartons, the stand-up pouch and the blister lidding.
Before I start detailing the designs, be aware of the following: The international standard ISO8317: 2003 describes the tests of resistance to child opening and ease of opening by adults. Children, 80% of a panel aged 42-51 months, must be unable to open the packaging in ten minutes, even after a demonstration, and 90% of a panel of 50-70 year old adults have to open and properly reclose the packaging in one minute. PPPA in the USA has similar directions.

In short we can say that a child resistant packaging usually requires a special ‘trick’ to open it. Something too complicated for most young children to work out, but easy to handle by seniors.
CR-slider for flexible packaging
Presto Products Company’s launch of a child-resistant flexible pouch opens up new opportunities for flexible packaging in markets where regulated products were previously only available in rigid packaging.
The Child-Guard technology leverages flexible packaging as a format meeting child-resistant mandates. The flexible pouch is child-resistant on the initial opening and subsequent re-openings using the Press-To-Engage (PTE) resealable slider.
 The slider is challenging for children under five years old, but simple for adult consumers, including seniors, noted the company. The technology allows brand owners to choose a flexible-packaging format as a lighter-weight alternative to rigid packaging and still meet child-resistant mandates.
The slider is challenging for children under five years old, but simple for adult consumers, including seniors, noted the company. The technology allows brand owners to choose a flexible-packaging format as a lighter-weight alternative to rigid packaging and still meet child-resistant mandates.
The Child-Guard slider meets Title 16 CFR 1700 of the Poison Prevention Packaging Act (PPPA) with ASTM D3475 classification from ASTM International.
The CR method is simple, effective and ingenious: As shown in the illustration, the slider opens with an easy “point, press and pull” motion. Users align the slider tongue with the groove, press down lightly on the slider’s top side and pull the slider across the zipper track for a smooth opening experience. The company claims that adult consumers, including seniors, will find the zipper simple to open, but it will be extremely challenging for children aged five and under addressed by the PPPA regulation.
Locked4Kids
Locked4Kids is a recloseable, child-resistant folding carton for packaging pharmaceuticals. The Locked4Kids carton consists of tear-resistant laminated paperboard and features a polyethylene terephthalate blister tray that locks in place. The special blister tray has small hooks on the long sides. These hooks are placed diagonally to one another, so that the tray is locked securely when it is pushed right in.
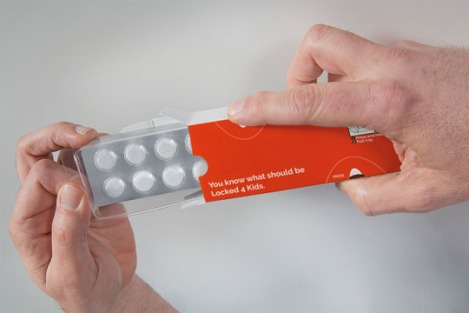
To remove the tray, both hooks have to be released simultaneously by pressing on the box firmly and evenly at the marked points with the thumb and forefinger. This is no problem for adults but impossible for children, whose hands are too small to span the width of the carton and push both hooks in at once, claims the inventor, Dutch company Ecobliss.
As a further deterrent, both sides of the box are coated with a tear resistant laminate to prevent children from simply ripping it apart.
Available in various sizes, the pack runs on conventional cartoning machines at standard speeds.
The CR-design developed by Ecobliss and marketed by Locked4Kids was recognized as the Best Innovation in Packaging in the Pharma Awards competition at CPhI Worldwide in October 2014 in Paris.

Ecobliss manufactured, the Locked4Kids carton with a special Romaco Promatic P 91S intermittent motion cartoner to package the Locked4Kids blister trays in laminated folding cartons.
The asymmetrical hooks attached to the blister trays mean particularly careful feeding is called for. Owing to their unusual geometry, the trays have to be additionally stabilised while they are being positioned in the cartons. The special feed tunnels used for this purpose result in highly precise processes. The Romaco Promatic P 91S achieves a maximum output of 80 Locked4Kids packs a minute.
The laminated cartons are removed and opened using Romaco Promatic’s positive carton opening system. The necessary vacuum is produced by low-noise Venturi nozzles. Promatic systems allow format changes to be completed in less than 15 minutes. The complete carton magazine is retractable to allow access to all cartoner components and speed up retooling.
MedLock EZ
We have another CR-folding carton for a blister card. The Medlock EZ compliance packaging from Colbert Packaging, consists of an outer paperboard shell housing a pill blister card that is locked into place with an integrated, patent-pending locking mechanism. The lock disengages by squeezing and holding touch-points at one end of the paperboard shell while sliding the blister card out through the other end. Once the blister card is exposed, medication can be pressed through one of the individual blisters. The patient can slide out the blister card to access a dose but cannot completely remove it. Sliding the blister card back into the outer shell reengages the lock.
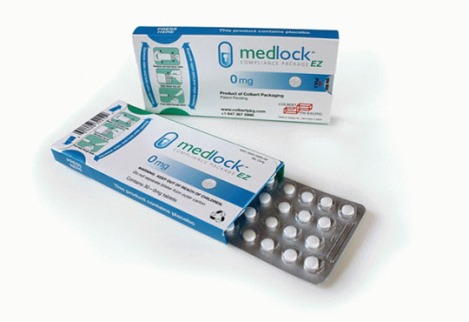
Highly versatile and customizable, the MedLock EZ delivers significant branding and sustainability benefits due to its paperboard-based design. The large printable area of the packaging can be used for branding as well as medication details, dosing instructions, dates and other information that engages patients and communicates a clear and consistent treatment regimen.

MedLock EZ will typically be made from uncoated solid bleached sulphate (SBS) paperboard, but it can also be made using other types of paperboard, and it can integrate various coatings and special effects.
Subjected to the testing protocols of the Consumer Product Safety Commission, the MedLock EZ has tested to F=1 for child-resistant and senior-friendly effectiveness.
Child-resistant push-through blister lidding
Amcor Flexibles introduced the Amcor Opening Feature (AOF) for child-resistant blister lidding to complement its product portfolio in pharmaceutical packaging. AOF allows users to access blister contents with a “targeted” push-through motion rather than the traditional peel or peel-push method.

The senior-friendly qualities and child-resistance of AOF Blister Lidding were confirmed through an independent study in accordance with the requirements of 16 CFR Part 1700. Blisters were produced using AOF blister lidding and the packaging met the test requirements of F=1 (only two children out of 50 were able to access one cavity). In addition, the Senior Use Effectiveness (SAUE) score was 98% compared to the normal target minimum of 90%, the company reports.
According to Amcor, AOF allows the use of a 2-ply structure compared with a standard 3-ply. The product contact layer remains the same as that of existing Amcor blister lidding products, helping users to streamline testing.
Without the need for peel tabs, total packaging sizes can be reduced up to 40% versus traditional peel-push and peelable blister cards, the company claims. The reduction reportedly does not compromise the barrier layer or the seal integrity of the packaging.

In my next article about CR-packaging I will describe 3 retrofit CR-solutions for aerosols and spray bottles. I discovered them when I did some research in the patents data bank, where a wealth of CR-designs is stored. The problem is you have to find your way.

Hi, Origin Pharma packaging have just released a new tamper evident, child resistant carton for blister pack strips.
You always can send me the techn. info and I will have a look.
Hi Thanks for getting back to me. You can reach out to them for any more details, they have produced some white papers. Origin pharma packaging have also produced a pp28 child resitant closure.
regards lee
https://www.originltd.com/pharma-packaging-innovations/smartx-child-resistant-tamper-evident-blister-pack-wallet